AI-Powered Clinical Trial Management & Documentation Solutions
CluePoints ensures that all clinical trial risk-related activities are recorded accurately and automatically within a centralized management platform equipped with AI-powered solutions leveraging advanced statistics and machine learning. Leveraging clinical trial documentation with artificial intelligence, users rely on RBQM for immediate risk review and action, mitigating issues and securing the safety and efficacy of clinical trials.
Risk Assessment & Mitigation
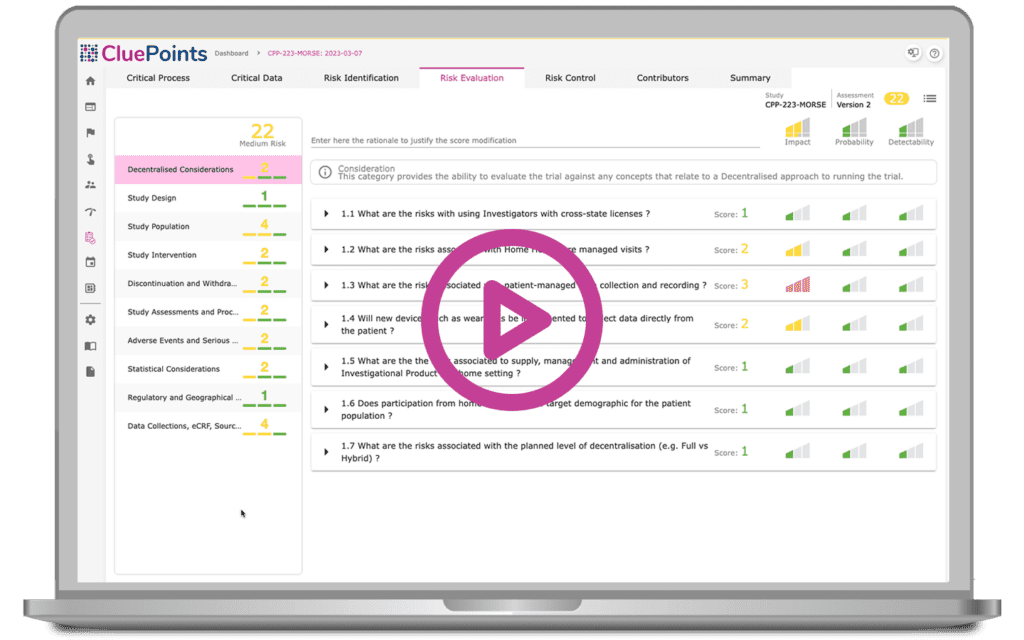
Risk assessments help users identify vulnerable clinical trial areas, define risk controls, and monitor research capabilities to detect evolving threat levels during study execution. Equipped with a complete audit trail, CluePoints’ dynamic, versioned risk assessments automatically capture stakeholder input, allowing users to define optimal risk control strategies, identify key indicators, and set risk thresholds. Our comprehensive management platform contains risk evaluation questionnaires and templates for users to easily customize and document the entire clinical risk assessment process for filing in the Trial Master File (TMF).
Signal & Action Tracker
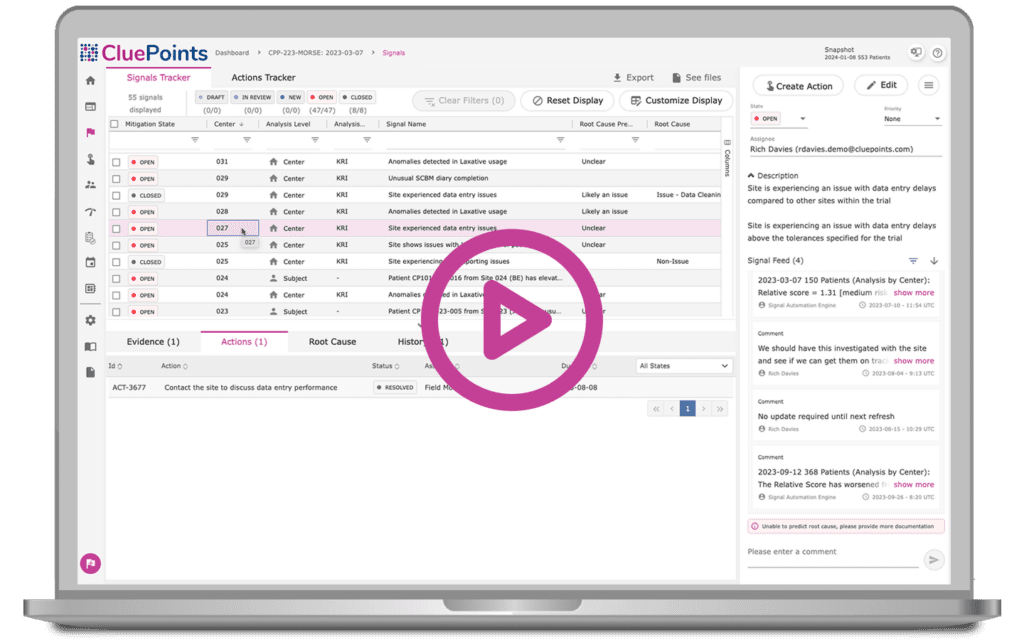
Our Signal and Action Tracker model helps assess issues identified through Central Statistical Monitoring. Integrated action management documents steps within a unified issue resolution management system, covering patent, research site, and country regulation concerns. Users can monitor risk signals and actions by status, annotate and document commentary throughout signal processing, deploy automatic auditing, and quickly share all risk mitigation activities with extensive exporting capabilities.
- Manage all potential issues detected within our platform or externally with a Clinical Trial Management System (CTMS)
On-Demand Demos
Dive into the full potential of CluePoints’ AI-powered RBQM solutions right from the comfort of your screen.