Exploring The Role Of AI And Machine Learning In Clinical Trials

We live in a rapidly evolving digital era, driven by an ongoing wave of groundbreaking technological innovations. Clinical research is no exception. In recent years, the industry has increasingly embraced technology to manage trials with greater efficiency, precision, and creativity. While many advancements have contributed to this transformation, two stand out as key drivers of […]
Centralized Monitoring – How To Unlock The Treasure Chest Of Analytical Insights
Chief Scientific Officer of CluePoints, Steve Young, discusses his thoughts on how Centralized monitoring has started to take off in the clinical trials industry even though it is still a fairly new trend. Where Does Centralized Monitoring Fit in Clinical Trials? Centralized Monitoring offers a way of remotely assessing all variables deemed to be indicative […]
Applying Risk-Based Quality Management To Investigator-Led Trials
Medical science is advancing at a never before seen rate, but costs are spiralling meaning clinical trials need to change if they are to ensure innovative treatments continue to be developed and delivered. Investigator-led trials are a vital part of the jigsaw of modern medicine. They can provide further evidence on novel products and devices, […]
From Risk-Based Monitoring To Risk-Based Quality Management
TL;DR Summary: Here are key definitions from our blog, “From Risk-Based Monitoring (RBM) to Risk-Based Quality Management (RBQM).” Risk-Based Monitoring (RBM): A strategy that uses software, data, and analytics to prioritize oversight on the most critical trial data and processes. RBM improves data quality, enhances patient safety, and reduces costs by enabling sponsors to identify […]
Webinar: The Future Of Clinical Trial Monitoring

In the wake of COVID-19, adoption of decentralized clinical research is taking off as sponsors explore ways to maintain continuity for existing studies while looking toward a more patient-centric future — bringing research to patients where they are with virtual trials. Regulators agree as reinforced by Stephen Hahn, FDA Commissioner, sharing that the agency will […]
New Paper: From RBM To RBQM – The Future Of Risk-Based Trial Management
A review of FDA marketing submissions between 2000 and 2012 revealed roughly one-third (32%) of all first-cycle review failures (16% of submissions overall) were driven by quality issues.1 Why was this? While the pharmaceutical industry had enjoyed economic growth in the 1990’s, drug makers started to face growing pressure at the turn of the century from […]
RBQM Doesn’t Have To Be Complicated, Risky Or Expensive: Q&A
On Wednesday, June 24, 2020, CluePoints delivered a webinar to demonstrate how using a combination of powerful analytics and comprehensive data visualization can be employed to ensure that no stone is left unturned in determining what issues are evident in studies and how to course-correct for a successful submission. If you missed the webinar, you […]
Beyond COVID-19: The New Normal In Risk-Based Monitoring (RBM) And Quality Management (RBQM)
Join our next webinar to learn first-hand how the CluePoints and Parexel partnership is driving best-in-class Risk-Based Quality Management Strategies to support clinical development. This session will focus on how early risk detection can be leveraged to detect issues across your trial and ensure operational excellence. As the COVID-19 pandemic continues, actions need to be prioritized to […]
Early Risk Detection During The COVID-19 Crisis – Your Questions Answered
On Tuesday, March 31, 2020, CluePoints delivered a webinar centred around the critical importance of early risk detection. Our presenter also shared insights on how to navigate risk management and risk assessment during the COVID-19 crisis. If you missed the webinar, you can watch it on-demand. Watching the webinar before digesting the answers shared in this document […]
Complimentary COVID-19 Risk Assessment Package Available To All At No Cost
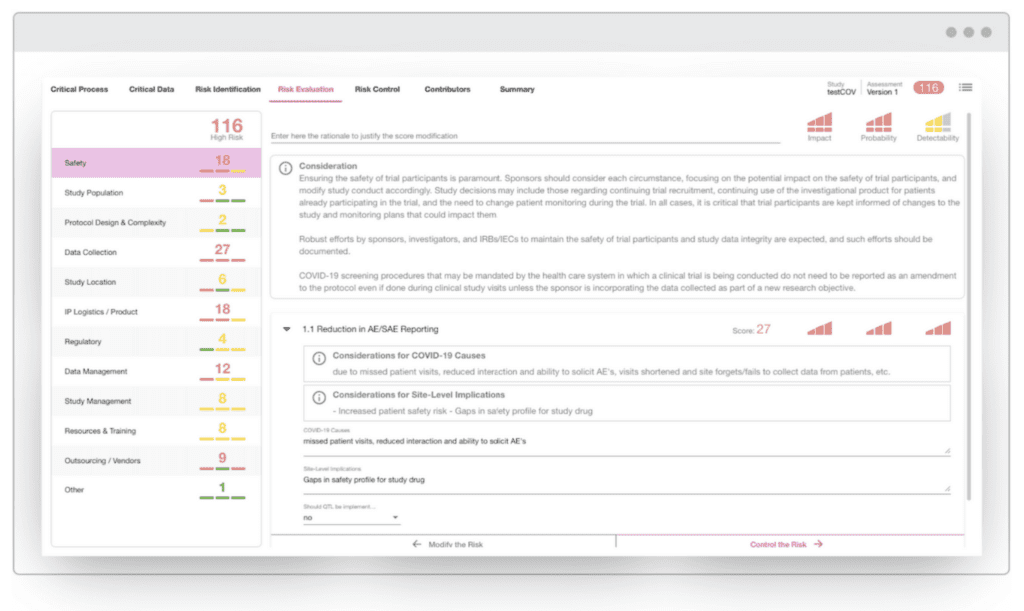
COVID-19 Risk Planning As advised in the coronavirus guidances recently issued by FDA, EMA, and MHRA, it is important to perform a fresh risk assessment for each study to identify and mitigate risks pertinent to the COVID-19 crisis. Your organization is likely already aware of key challenges that will be common to most if not […]

