As guidance for Industry has evolved, ICH E6 (R2) for example, presenting new recommendations for ensuring patient safety, organizations have faced significant challenges in translating the guidance into tangible operating practices to satisfy the requirement of an entirely Risk-Based Approach to Study Execution (RBx). Being the leader in Risk-Based Quality Management Solutions, we’ve garnered a wealth of experience in the space, perfectly positioning us to offer support in preparing for site inspections and implementing Risk-Based Monitoring (RBM)/Risk-Based Study Execution (RBx).
SERVICES
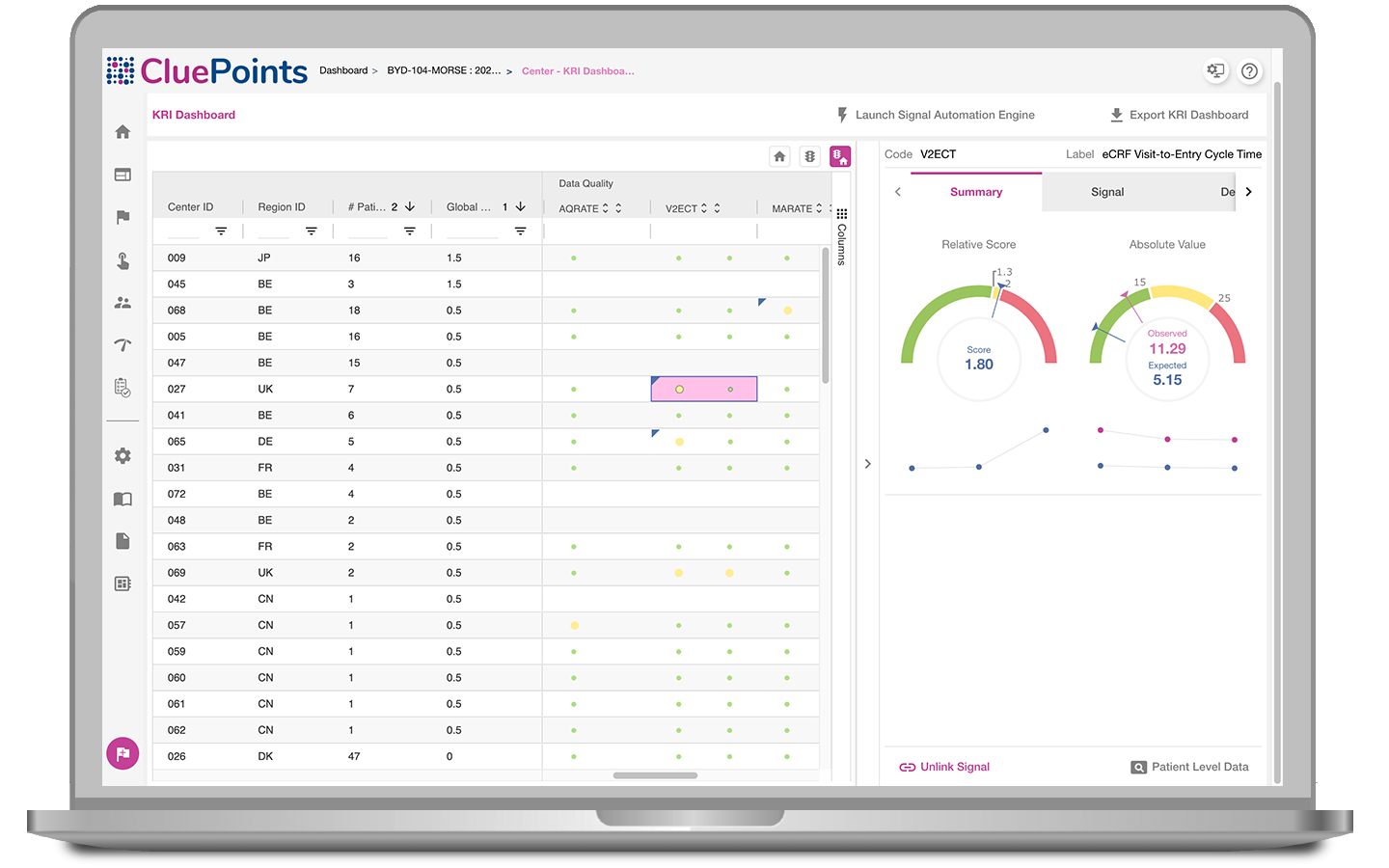
Central Monitoring
CluePoints can support your proposed or ongoing clinical trial by providing the platform as a service. Our industry expert Data Analysts will configure the system on your behalf before processing your clinical and operational data to report the findings back to you. Let CluePoints inform you as to where to focus your operational activities to maximise data quality and ensure overall trial success.
Services
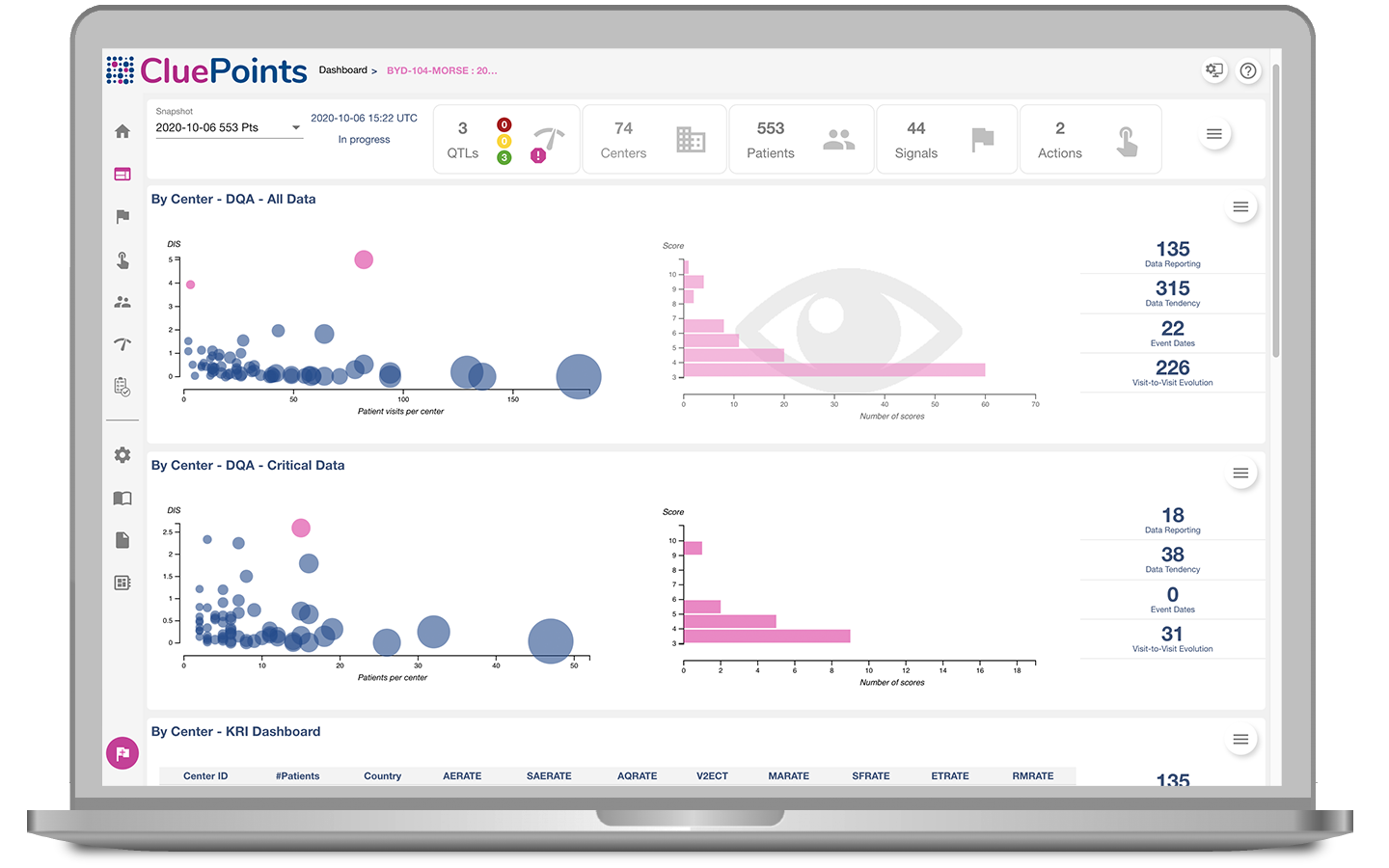
RBM/RBx Implementation
CluePoints is the industry leader in RBM/RBx implementation and our professional services team are available to guide your journey to implementation success. Whether that is helping define your holistic RBx strategy, aligning with ICH E6 (R2) guidance, or simply helping define tactical monitoring approaches and KRIs, the CluePoints professional services team have the historical experience to support you.
Services
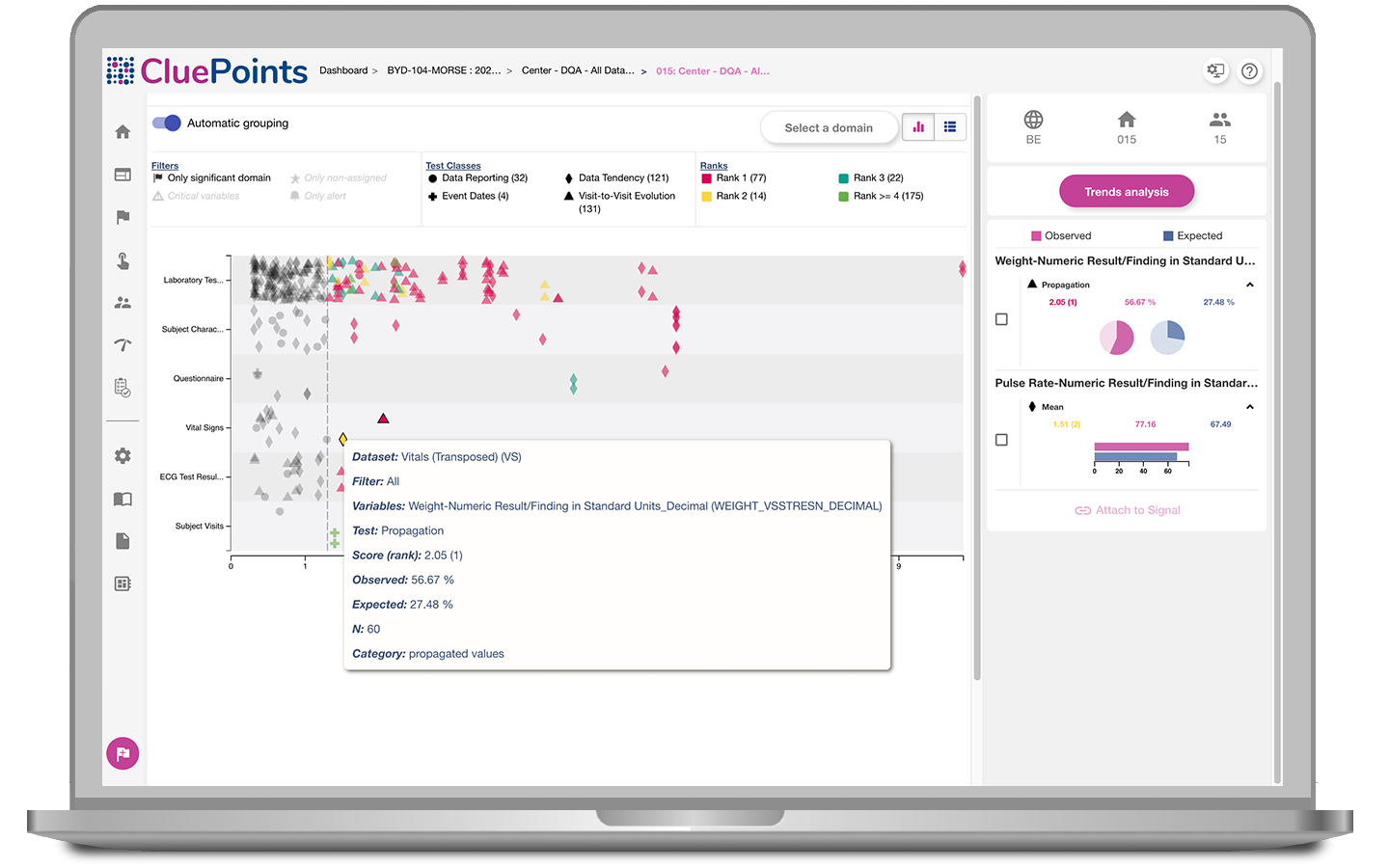
Site Inspection Readiness
Your trial may be complete, but do you have full visibility on site performance and the quality of the data supplied? Will any of your sites stand out when your data is reviewed by the regulators, leading to a site inspection? Our Site Inspection Readiness service can interrogate all of your clinical and operational data to identify sites at risk of inspection, helping you proactively prepare in advance
Services
Mergers & Acquisitions
Does the clinical data that supports your proposed acquisition contain underlying issues that will compromise its ability to eventually support a successful regulatory submission? CluePoints can test the available data to look for atypical patterns, or absence thereof which help verify the robustness of the data and associated processes.

Got a Question?
Whether you’re looking to get started or need support, we’re here to help!
Featured Resource
Tufts CSDD Impact Report
This study takes a proactive approach to understanding the dynamics between the enthusiasm around and adoption of RBQM, designed to enhance clinical trial quality and efficiency.
Featured Webinar
RBQM Adoption & ICH E6(R3) Updates
This recorded webinar dissects the Tufts CSDD Impact Report as well as new clinical trial execution models and ICH E6(R3) regulatory guidance updates.
